Cancer Prognosis
Cancer Immune Checkpoint Blockade Prognostic
Novel Immune Checkpoint Blockade PD-L1 Quantification
Cancer immunotherapy treatment has made great strides in the treatment of various cancers, specifically by targeting the immune checkpoint blockade. This blockade helps regulate programmed cell death and are vital targets during cancer treatment. Unfortunately, these treatments are not always successful, as not all cancers and cancer cells present with the necessary checkpoint ligands. Researchers at Virginia Commonwealth University have developed a novel diagnostic that can determine the level of immune checkpoint blockade ligand on tumor exosomes from systemic samples allowing for the prediction of responsiveness to cancer immunotherapy.
The technology
This novel assay detects immune blockade ligands, specifically programmed death-ligand 1 (PD-L1), on tumor exosomes. PD-L1 levels on the tumor exosome has been shown to be predictive of the immune checkpoint blockade response to immunotherapy. Detection of PD-L1 is achieved with an exosome-hybridization chain reaction (ExoHCR) as shown in Figure 1. This technique has significant advantages over the current technique for quantifying ligand levels, which necessitates a biopsy followed by immunohistochemical analysis. This method is much more rapid than current diagnostics as the HCR is spontaneous, enzyme-free, and isothermal. In addition, the ExoHCR diagnostic minimizes invasiveness, as it can be performed from blood samples rather than tumor biopsies. Furthermore, errors from tumor heterogeneity that are common in tissue biopsies are minimized allowing increases specificity and sensitivity. This technology is also highly adaptable. By manipulating the targeting antibody this diagnostic can be tailored to various cancer biomarkers and immune checkpoint ligands.
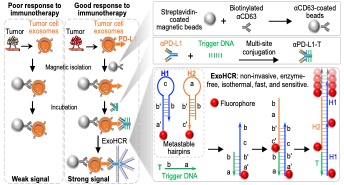
Figure 1. Schematic illustration of the ExoHCR diagnostic used to profile PD-L1 tumor cell exosomes.
