Small Molecule
Novel cGAS Therapeutic
Small molecular degrader of Cyclic GMP-AMP synthase
Cyclic GMP-AMP synthase (cGAS) is a cytosolic DNA sensor that elicit type-I interferon (IFN-I) immune responses in the presence of cytosolic DNA. In the events such as nuclease malfunctions, self-DNA accumulates in the cytosol and causes cGAS overactivation, which can further lead to autoimmune diseases. Unfortunately, current strategies to inhibit cGAS have had mixed results, with numerous off target effects and limited efficacy. Researchers at Virginia Commonwealth University (VCU) have developed a small molecule that allows for the targeted and long-lasting protein degradation of cGAS for the treatment of cGAS overactivation-related diseases.
The technology
This novel small molecule is a molecule-based protein degrader or cGAS-specific proteolysis targeting chimera (PROTAC). This small molecule works by facilitating the targeted binding of ubiquitin activating enzymes and ubiquitin ligases to the cGAS molecule. This then results in the overall ubiquitination and proteolysis of the cGAS molecule and therefore less activation of type-I interferon immune response shown in Figure 1. The targeted nature of this PROTAC allows for selective degradation of cGAS with a high efficacy while minimizing adverse effects typical of other cGAS inhibitors. Further, because this molecule triggers degradation and not inhibition, treatment with this molecule causes a long-lasting downregulation of the cGAS and the downstream immune response.
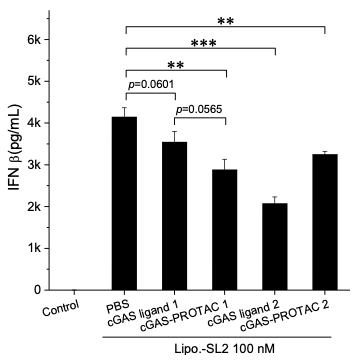
Figure 1. IFN β concentration in RAW264.7 macrophage cells after treated with different drugs for 10 h. The drugs were co-added with 100 nM DNA-loaded lipofectamine 2000. **p<0.01, ***p<0.001. The concentrations of PF-06928215 and cGAS-PROTAC 1 were 4 μM; RU.521 and cGAS-PROTAC 2 were 1 μM.
