Research tools
Generation of StarD5 Knockout Mice and uses thereof
StarD5 is a cholesterol binding protein believed to be involved in the intracellular translocation of cholesterol. An increase in levels of StarD5 is associated with increased levels of cholesterol in the plasma. High levels of cholesterol lead to increased blood pressure, and increased risk of heart disease, heart attack and stroke.
The technology
Researchers at Virginia Commonwealth University have developed a generation of StarD5 knockout mice which do not express the StarD5 gene through RNA sequencing and crossing of heterozygous mice. Testing confirms the correct sequencing showing positive results.
To create a starD5 knockout mouse, a guide RNA was designed to create a double-strand cleavage upstream of the ATG start site. Through a translational frame-shift and mutations, sequences were initiated for efficient translational initiation as well as a site for screening purposes. Seventeen pups were obtained, screened and sequenced. Heterozygous male and female mice were crossed to generate homozygous StarD5 knockout mice. Subsequent sequencing confirmed correct targeting. A western analysis of the liver and peritoneal macrophages (Figure 1) showed no StarD5 protein present, thus showing successful elimination of this protein in the animal. These results could lead to important pathological insight into cholesterol as well as pharmacological tools to control related conditions.
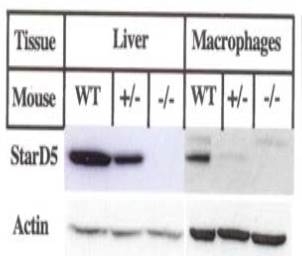
Figure 1. Western blot analysis of StarD5 knockout mice. Total protein extracts were prepared from liver and peritoneal macrophages as indicated and used to perform western blots for StarD5 and actin as a control.
